
COMPAR-EU is starting its fourth year, and now, after completing the network meta-analysis to estimate the relative merit of self-management interventions, we are also working to facilitate the use of this new knowledge the project is gradually delivering. One of the key steps of COMPAR-EU is the development of different outputs to facilitate decision-making for different stakeholders and scenarios. The goal is to produce interactive tools for health care professionals, patients, and policy makers, among others.
The tools that are being developed are Summary of Findings (SoF) tables, Evidence to Decision (EtD) frameworks, and decision aids. These tools were originally developed by the GRADE working group in the context of a European project called DECIDE. They are part of the GRADE approach, which is a systematic approach to assess and communicate the certainty of evidence (“quality of evidence” or “confidence in an estimate of effect”) and the strength of recommendations. COMPAR-EU uses GRADE to evaluate the certainty of evidence about the effects and will also use it to formulate recommendations.
Summary of Findings tables (SoF)
These tables present the main findings of a review in a transparent, structured, and simple tabular format. In particular, they provide key information concerning the certainty or quality of evidence (i.e., the confidence or certainty in the range of an effect estimate or an association), the magnitude of the effects of the interventions examined, and the sum of available data on the main outcomes.

The SoF tables will be interactive to improve understanding and use of evidence of the effects of healthcare interventions. They allow users to interact with the presentation by viewing more or fewer outcomes, more or less information about each outcome, information about the absolute effects as numbers, words or graphs, cursor over explanations and links to more detailed explanations of basic concepts (e.g., “95% CI”).
Evidence to Decision (EtD) frameworks
EtD frameworks help panels use evidence in a structured and transparent way to inform decisions in the context of clinical recommendations, coverage decisions, and health system or public health recommendations and decisions. EtD frameworks have a common structure that includes the formulation of the question, an assessment of the evidence, and drawing conclusions, though there are some differences between frameworks for each type of decision. EtD frameworks inform users about the judgments that were made and the evidence supporting those judgments by making the basis for decisions transparent to target audiences.
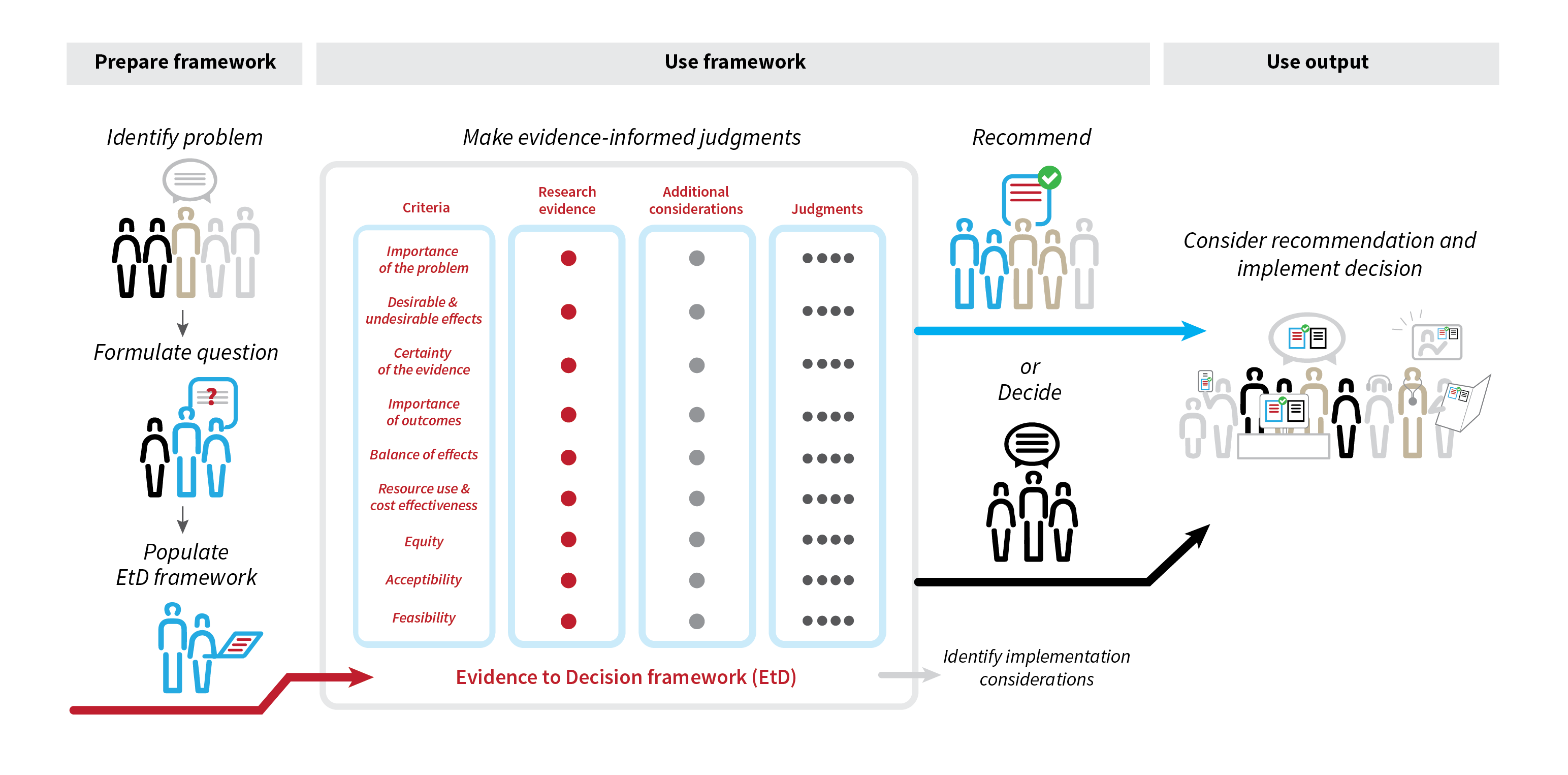
Illustration: Sarah Rosenbaum, Norwegian Institute of Public Health
The frameworks take into account the magnitude of desirable and undesirable effects, stakeholder views on the importance of different outcomes, information on resource use and cost-effectiveness, impact on equity, and other aspects like acceptability or feasibility of the interventions. COMPAR-EU partners are producing the different pieces of evidence that will populate the frameworks; comparative effectiveness analysis via network metanalysis, cost-effectiveness evidence, and contextual factors evidence, helping formulation of recommendations and future implementation of successful self-management interventions.
Using the frameworks, COMPAR-EU panels are being assembled and will formulate clinical recommendations using a population perspective. These recommendations could be then applied (adopted) or adapted to different settings. The frameworks will be particularly useful for decision makers in different settings, as they will be able to review the evidence and the judgments made by the panel, and decide whether to adopt the recommendation (use it directly) or to adapt to their particular setting.

Dr Pablo Alonso
Pablo is a senior researcher at the Iberoamerican Cochrane Centre (Biomedical Research Institute Sant Pau). His research is focused on systematic reviews and guideline methodology, with a special interest in incorporating the patients and the public perspective into health care decisions. He is responsible for the Barcelona GRADE center, and a Board member of the GIN and of the GRADE Guidance Group.
Patient decision aids
The project will also develop decision aids to enable comparisons between multiple potential options. The decision aids will include information on the most important outcomes, the certainty we can place on the results of the research results, as well as practical considerations about the self-management interventions being considered (e.g., burden). These tools will be designed for the use in face-to-face clinical encounters or to be used by patients in advance of a clinical encounter. The aids will be produced in six languages (English, French, German, Spanish, Dutch and Greek) using plain language.
All the tools will be available in a web-based platform that is being developed and will be made available in early 2022. The platform will include many other things, but this is something that will rather be revealed in another blog post.
